Electrolytes for Divalent Cation Batteries: Understanding and Design
The LEGEND project aims to design electrolytes for divalent cation-based batteries, specifically using Ca(II), Mg(II), and Zn(II)—abundant elements that offer high energy density. The rational design of high-performance electrolytes requires a deeper understanding of ion solvation and transport mechanisms, as well as structuring and deposition phenomena at electrode interfaces. This project leverages and develops molecular simulation methods and machine learning to build a library of high-potential electrolytes, which will be experimentally validated.
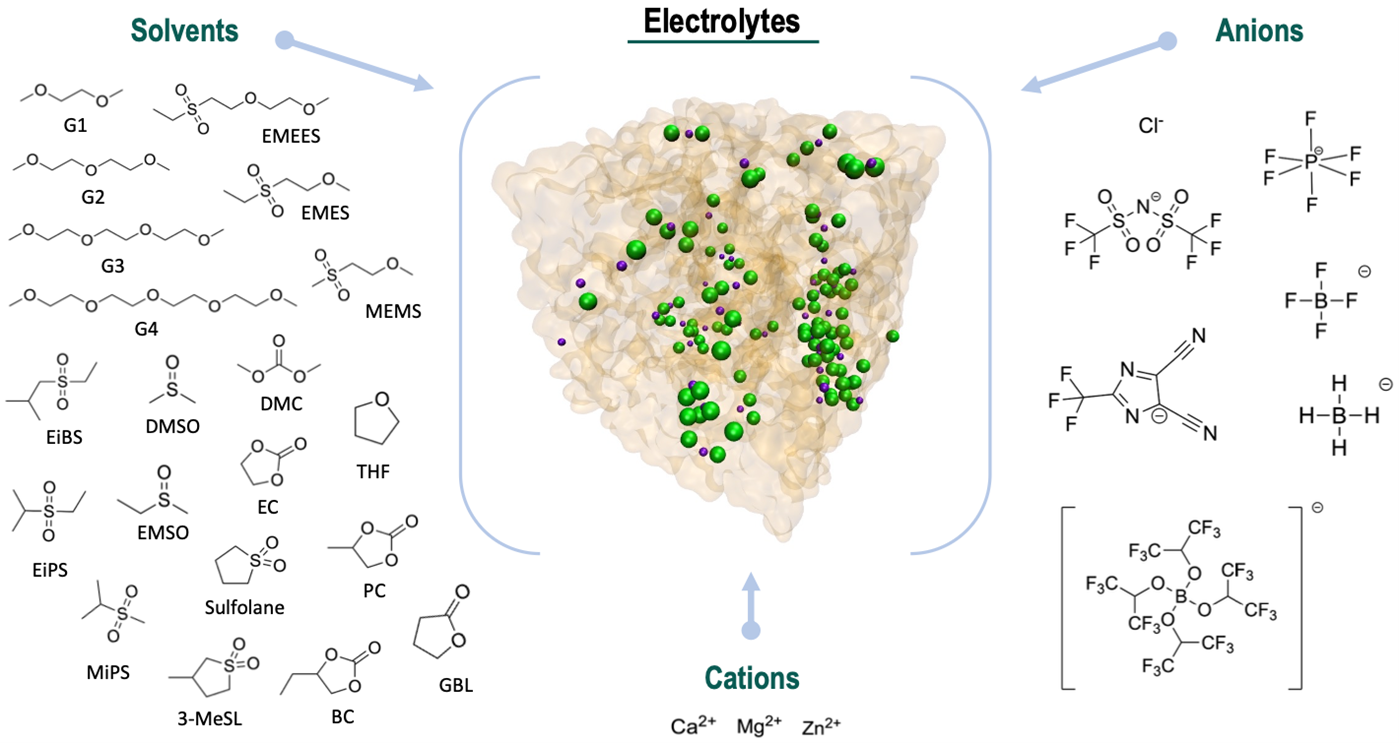
Following the development and refinement of polarizable molecular interaction models (force fields) for divalent cations and electrolyte components, a set of descriptors for ions in the electrolyte phase is being generated through high-performance computing. These descriptors include thermodynamic, structural (solvation shells, speciation), transport (diffusion, conductivity), and dielectric properties. Several dozen compounds are under investigation, including ethers (glymes), sulfones, carbonates, and various weakly coordinating anions. Trends in the relationships between molecular structure and properties are beginning to emerge.
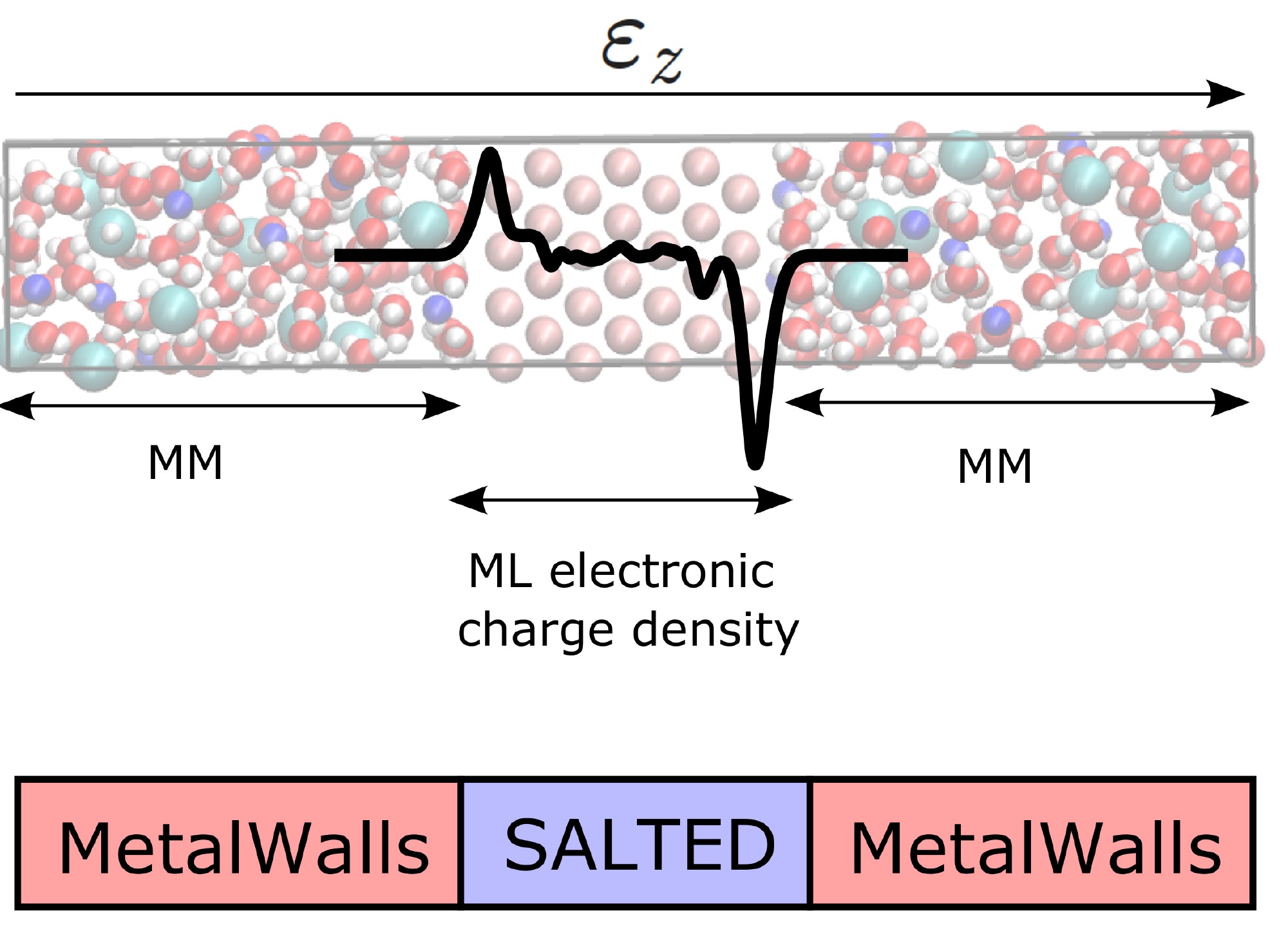
The modeling of interfaces, particularly the negative electrode in this project, is at the methodological development stage, with initial studies on model systems. Ab initio simulations (quantum chemistry) of the interface between metallic zinc and water have revealed the organization of molecules near the surface—critical information for understanding cation desolvation mechanisms. The SALTED model, which describes electrode polarization and was developed in the BATMAN project of the PEPR Batteries, has been adapted for the systems in this project. Concurrently, neural network potentials (NNPs) for interactions between the electrode and electrolyte are being developed.
The next steps of the project will expand the chemical space of electrolytes to include borate anions, ionic liquids, and consider the role of water as an impurity. For interface modeling, once the NNP training is complete, calcium-based electrolytes using glymes will be studied at the metal surface.
Autres Highlights