High‑energy‑density aqueous organic redox flow batteries
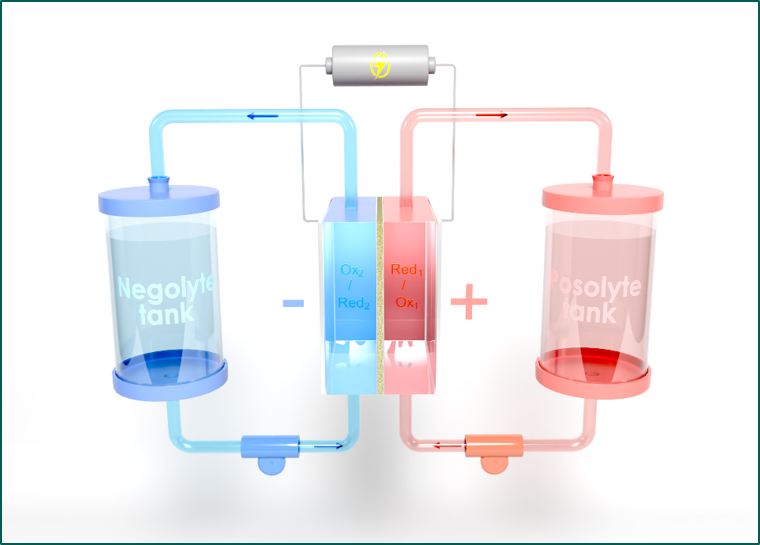
The Discovery project aims to increase the energy density of aqueous organic redox flow batteries (AORFBs), a promising technology for storing intermittent energy. To achieve this, insoluble redox species (boosters) are added to the reservoirs that can exchange electrons with soluble redox mediators in the aqueous electrolyte. For the battery to function properly, the redox potentials of the boosters and mediators must be similar, and the electron exchange reaction must be rapid and spontaneous.
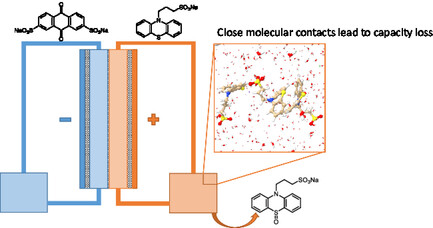
In this context, we undertook the synthesis of new mediators covering a wide range of redox potentials. The focus was on mediators for the positive electrode, which remain little studied, particularly those from the phenothiazine and ferrocene families. Some phenothiazine derivatives carrying solubilizing groups exhibit promising solubility and redox potential properties for the application. However, these compounds show a rapid loss of capacity in batteries, linked to a degradation mechanism involving a disproportionation reaction (Figure). These results were published in Chemistry Europe.
To limit this phenomenon, bulky solubilizing groups were introduced to prevent π-stacking dimerization, which is responsible for the disproportionation reaction.
In addition, a new family of ferrocene derivatives has been synthesized. These compounds feature solubilizing chains of varying chemical nature and length. Physicochemical studies of these molecules allowed the identification of the most promising architectures for the intended application and highlighted original solubilizing groups that have not yet been explored for AORFBs. Disubstituted derivatives were also prepared, providing an additional lever for adjusting the redox potential. These efforts led to an invention disclosure. In parallel, exploratory research on manganese complexes was initiated this year.
The project is also focusing on the development of negolytes. Work began this year with the goal of developing a patented synthesis process (WO 2024/261060) for naphthoquinones (ACS Applied Materials & Interfaces) and the preparation of azaanthraquinones, a family that remains little explored for this type of application.
Regarding boosters, polymers incorporating redox motifs directly into their structure have been synthesized and electrochemically characterized. Perylene diimide–based polymers linked by glycol ether bonds show the required properties for use as boosters: redox potentials compatible with certain mediators and good processability as inks. The development of pendant polymers, in which a polymer backbone is functionalized with redox entities, is currently underway.
Finally, the membrane, a key component of redox flow batteries, is also being developed. Membranes of varying thicknesses have been obtained from sulfonated alkyl-aromatic polymers synthesized via Friedel–Crafts polyalkylation. These membranes exhibit excellent mechanical and chemical stability, as well as ionic conductivity close to that of Aquivion. Initial tests of amphoteric membranes, based on copolymers or polymers containing both anionic and cationic functional groups, have shown promising results.
Autres Highlights