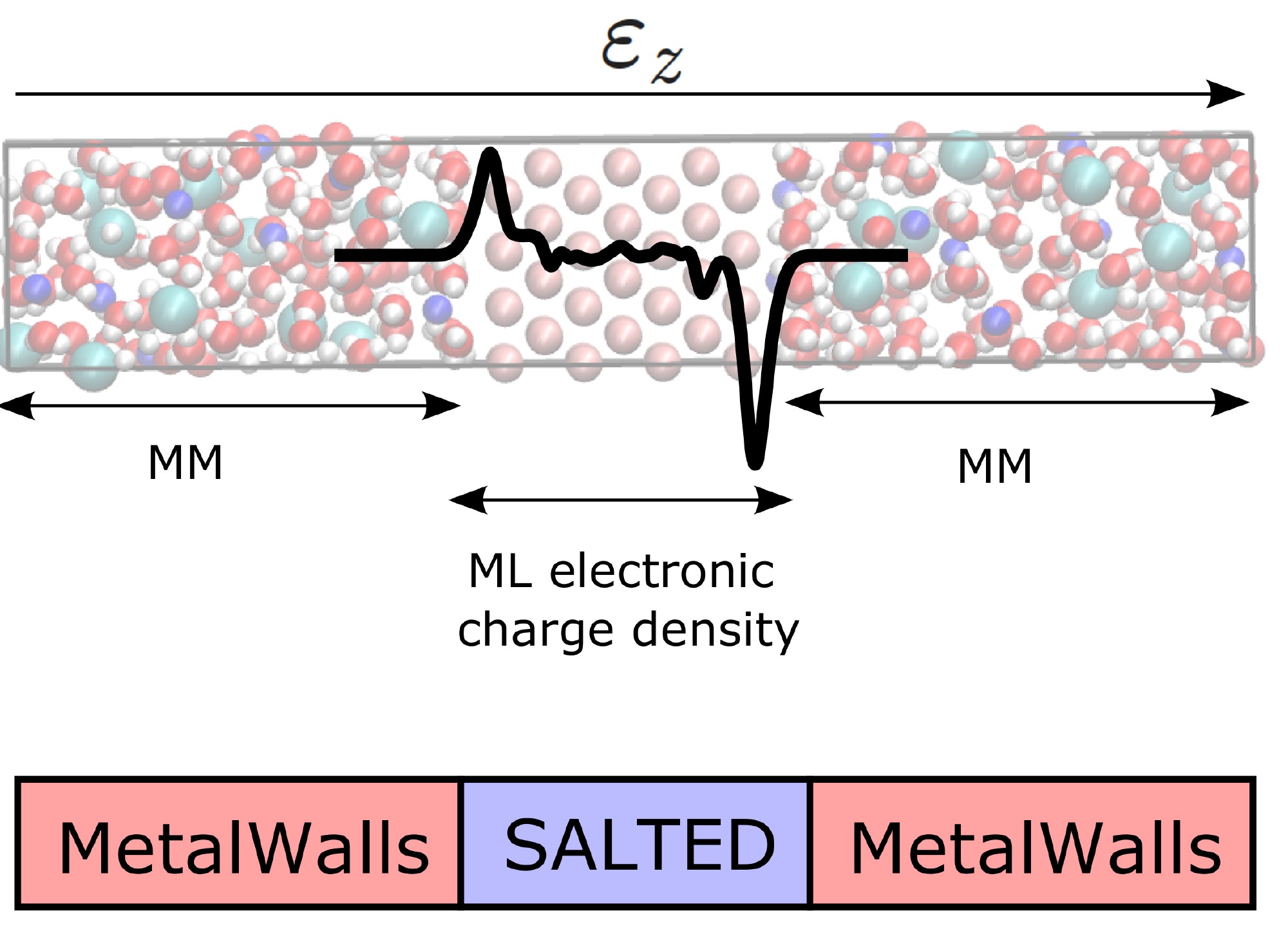
Latest Advances in All-Solid-State Lithium Metal Batteries
The LIMASSE project aims to develop and optimize materials, coatings, and processes to prepare all-solid-state cells using NMC/Li metal and Sulfur/Li metal chemistries, leveraging advanced characterization techniques. The latest advances of the LIMASSE project, with a focus on the development of materials and coatings, were presented at the PEPR Batteries Days on January 14 and 15, 2026.
A significant portion of the work focuses on cathodes, particularly through the synthesis of monolithic NMC811 particles (Figure 1). Research continues with the study of various coatings compatible with sulfur-based electrolytes. For the Sulfur/Li chemistry, the use of a Li₄PS₄I electrolyte, which exhibits better stability with lithium metal compared to the “classic” Li₆PS₅Cl (LPSCl), has enabled an increase in the cathode’s areal capacity. While initial capacity retention remains limited, these results are promising. The next phase of the study aims to further refine the composition and preparation of the cathode to improve the Sulfur/Carbon/Electrolyte interfaces.
Another aspect of the LIMASSE project involves the use of polymers for the preparation of hybrid or polymer electrolytes. In the case of hybrid electrolytes, the reactivity between Li₆PS₅Cl argyrodite and the PVDF-HFP polymer is being investigated. NMR studies have shown that LPSCl/PVDF-HFP mixtures do not exhibit changes in the ³¹P signal up to 200°C, although the formation of LiF at 200°C can be observed in the ¹⁹F signal. Cells made with a Li₆PS₅Cl separator containing 10 wt% PVDF-HFP do not appear to show degraded electrochemical performance. The other polymer-based approach focuses on synthesizing polymer electrolytes for Lithium-Sulfur chemistry. The strategy involves adding an anionic group to a polyethylene glycol-based polymer to repel polysulfides formed during sulfur lithiation. Initial results show an improvement in lithium transference number, but ionic conductivity is not better than the reference.

Autres Highlights