New Electrolyte for Fluoride-Ion Batteries
The FRISBI project aims to develop all-solid-state fluoride-ion batteries as an alternative to lithium-ion batteries, due to their high theoretical energy densities. The work focuses on designing solid electrolytes with high ionic conductivity, exploring fluorinated electrode materials, and integrating these components into electrochemical cells. Tin-based electrolytes showing promising ionic conduction are being studied, and various structural families of electrode materials are being explored.
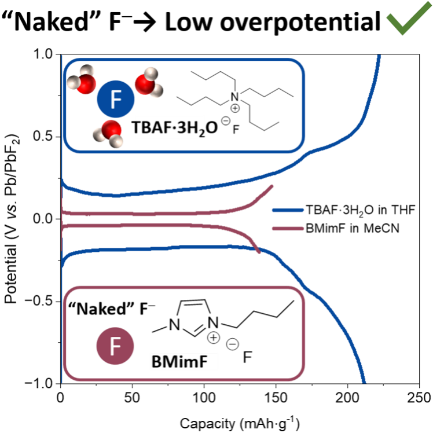
Fluoride-ion batteries rely on the migration of F⁻ ions between two electrodes. However, the development of fully solid-state systems remains severely limited by the lack of solid electrolytes capable of ensuring efficient fluoride ion transport at room temperature. In this context, the use of liquid electrolytes is a relevant approach for studying insertion electrode materials, as it avoids the limitations associated with solid–solid interfaces and provides more direct access to the intrinsic electrochemical properties of the electrodes. In this regard, a new electrolyte composed of 1-butyl-3-methylimidazolium fluoride (BMimF) dissolved in acetonitrile has been investigated. Unlike commonly used salts such as TBAF·3H₂O, in which the fluoride ion is strongly coordinated by three water molecules, BMimF yields much less solvated fluoride ions. Fluorine-19 NMR analyses show that this solution provides a significant fraction of “free” (“naked”) fluoride ions, promoting their mobility and electrochemical reactivity.
Electrochemical measurements reveal high ionic conductivity (15.3 mS·cm⁻¹ at room temperature) and an electrochemical stability window ranging from 0.37 to 1.59 V vs. Pb/PbF₂, comparable to that of the reference 1M TBAF electrolyte in THF. When tested in a symmetric Pb/PbF₂ cell, this electrolyte enables reversible cycling with a very low initial overpotential of 75 mV at 6 mA·g⁻¹, one of the lowest values reported for this system. When paired with a BiF₃ positive electrode, it also allows for reversible conversion reactions with moderate overpotential (≈160 mV), confirming the effectiveness of fluoride ion transport.
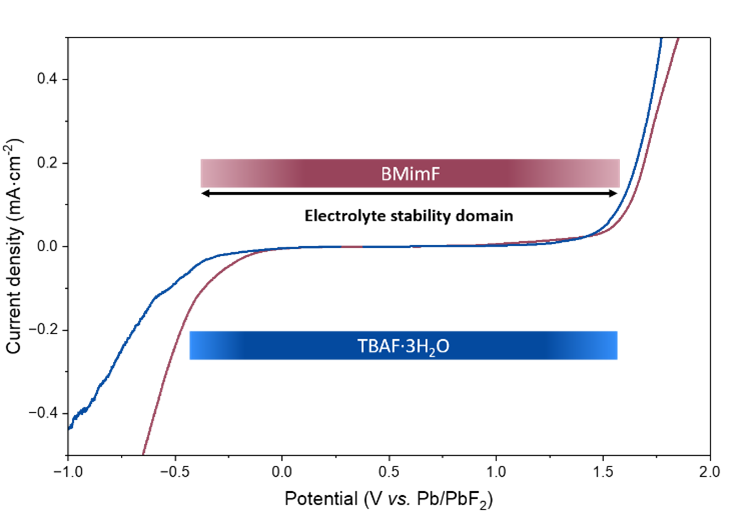
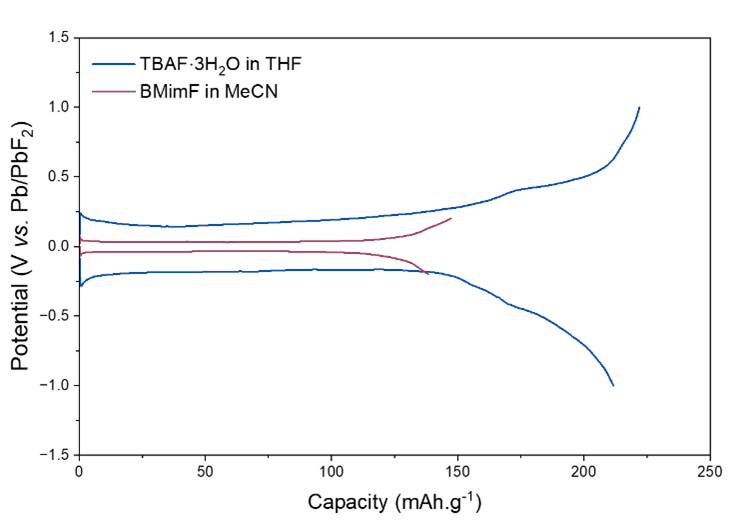
Figure 1: Electrochemical stability window of TBAF·3H₂O in THF and BMimF in CH₃CN on the left, and the first full cycle of symmetric Pb + PbF₂ + VGCF cells with TBAF·3H₂O in THF and BMimF in CH₃CN as the electrolyte.
Additionally, the chemical compatibility of this electrolyte was evaluated with the insertion material CsMnFeF₆. Unlike the reference hydrated electrolyte (1M TBAF in THF), the BMimF solution limits cation dissolution and preserves the structural integrity of the material after several days of contact. While the electrochemical performance in terms of insertion remains modest, these results demonstrate that fluorinated imidazolium salts represent a promising pathway for designing dry, conductive, and more stable electrolytes for next-generation fluoride-ion batteries.
Autres Highlights